Fujifilm developed a non-animal based recombinant peptide (RCP), taking advantage of the know-how from our photographic film past. RCP is a biomaterial, applicable in various forms such as sponges, granules and film for a broad range of medical and pharmaceutical applications.
Features
- No animal-derived components (xeno-free)
Our yeast-based manufacturing platform ensures the highest level of safety for the users of cellnest™. - Cell-adhesion capability equal to or exceeding animal collagen
cellnest™ contains multiple copies of RGD cell-adhesion motifs. - Biodegradable and bioabsorbable
cellnest™ is a perfect material for clinical research due to its excellent biodegradable and bioabsorbable properties. - Uniform size
cellnest™ can be processed to form a wide variety of structures. Low endotoxin (≤10 EU/g)
Innovative and Effective Structural Design
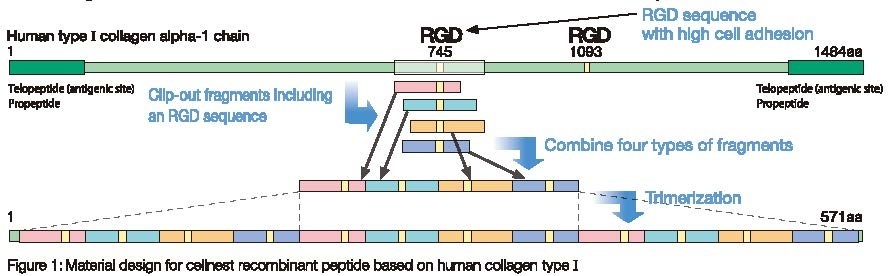
The α1 chain of human type I collagen contains RGD cell-adhesion sequences. To construct a highly adhesive collagen derivative, we first synthesized four DNA fragments corresponding to the RGD-containing peptides indicated in the figure. These fragments were then joined together to generate a core coding unit of the collagen derivative. The final DNA fragment consists of three tandem repeats of this core unit. The recombinant polypeptide (cellnest™) expressed from the final fragment lacks the antigenic telopeptides, while retaining the highly adhesive properties of collagen.
Versatile and Adaptable Biomaterial
cellnest™ is manufactured as a highly homogeneous peptide of approximately 51 kDa. The structural uniformity of this collagen-like molecule allows for its easy handling and enables its processing into diverse physical forms (such as sponges, films, granules, and porous particles).
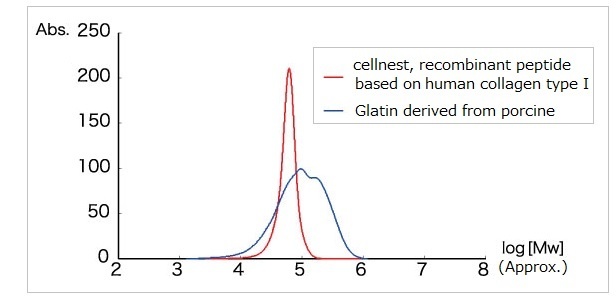
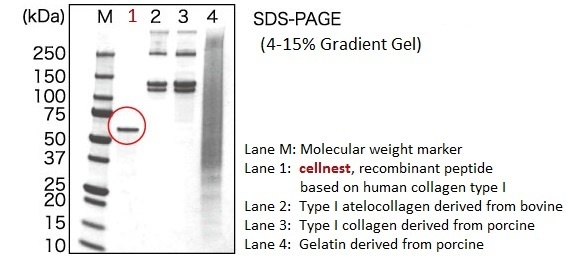

Consistent and Reliable Quality
cellnest™ is a recombinant peptide produced in yeast. Thus, it displays minimum lot-to-lot variability in performance. The use of a yeast expression system also guarantees the stable supply of this product.

Application Data
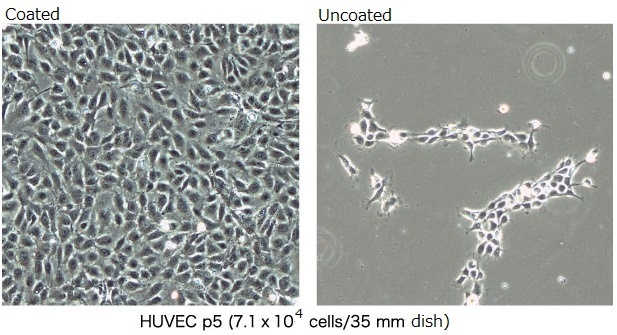
Human umbilical vein endothelial cells (HUVECs) were cultured on tissue culture dishes coated with cellnest™ (0.1%). We verified that this recombinant peptide improves the morphology of these human cells. We also confirmed that cells grown on cellnest™-coated dishes show significantly enhanced adhesion and spreading, as compared with cells cultured on uncoated dishes.
Validated Cell Types
Endothelial cells (HUVEC), keratinocytes (NHEK), myoblasts (C2C12), osteoblast-lineage cells (MC3T3-L1), fibroblast-like cells (MC3T3-E1), epithelial-lineage cells (CHO-K1, Vero, MDCK), and kidney cells(CV-1).
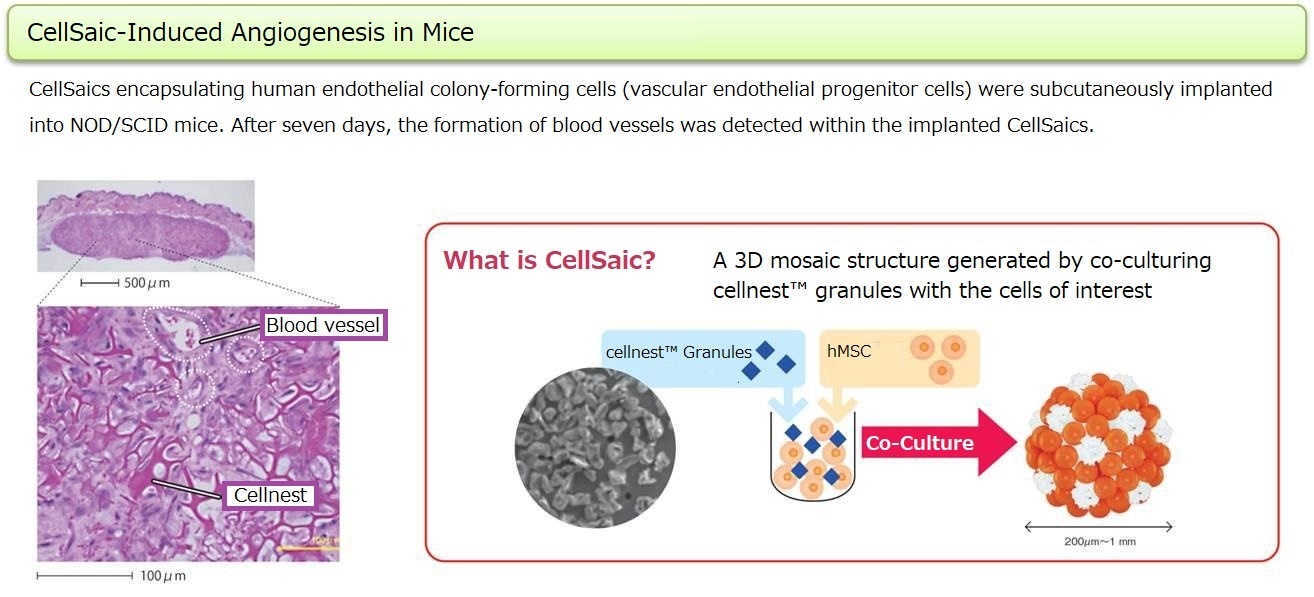
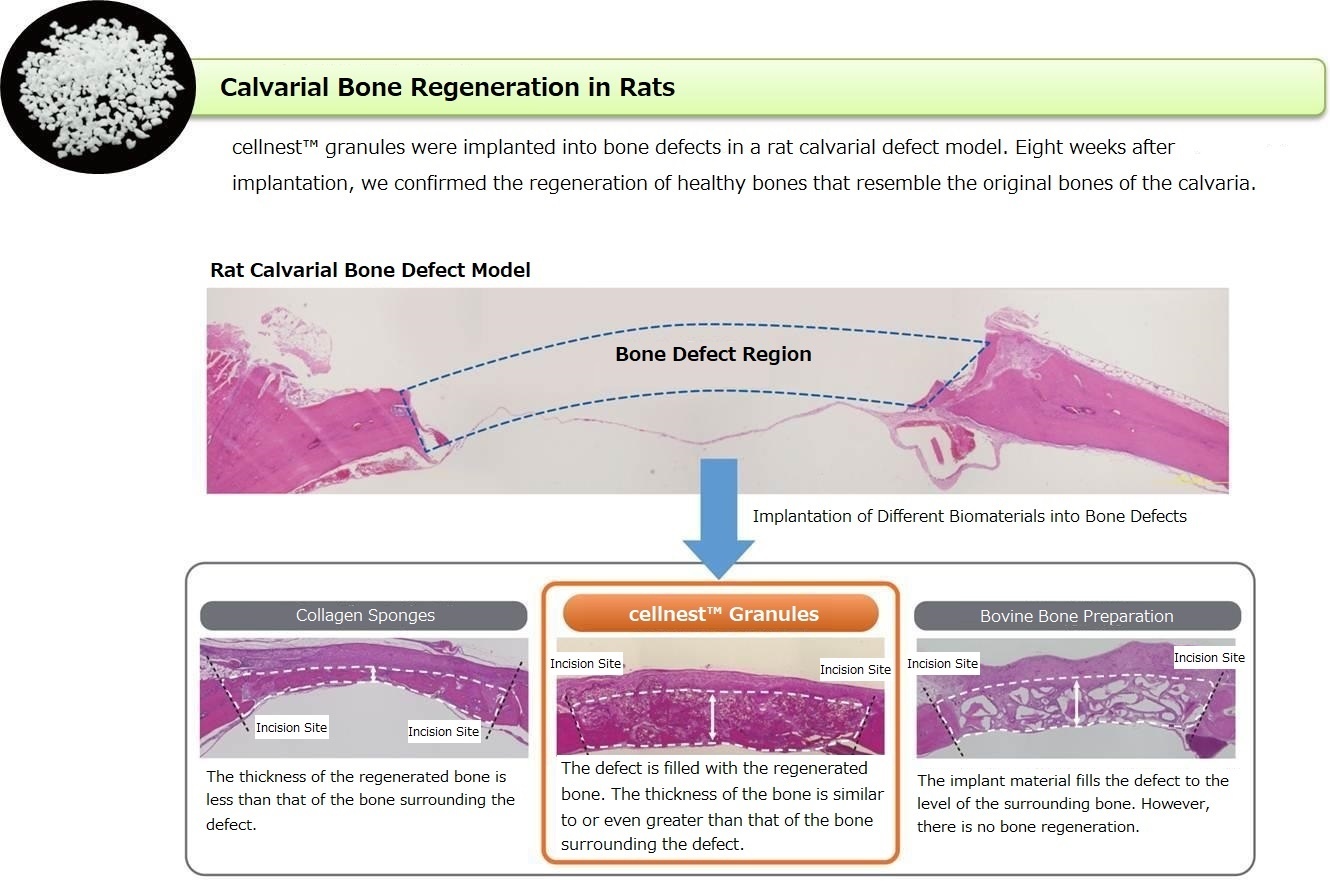
Protocols
References
Kentaro Nakamura, Yasuhiko Tabata.
A new fluorescent imaging of renal inflammation with RCP.
Journal of Controlled Release, Volume 148, Issue 3, Pages 351-358
Kentaro Nakamura, Reiko Iwazawa & Yasuhiro Yoshioka.
Introduction to a new cell transplantation platform via recombinant peptide petaloid pieces and its application to islet transplantation with mesenchymal stem cells.
Transplant International, Volume 29, Issue 9, September 2016, Pages 1039-1050
・Yasuhiro Yoshioka, Kentaro Nakamura & Reiko Iwazawa. BIO Clinica, Volume 31, Issue 3, 2016, Pages 62-66. (in Japanese)
・Hirohiko Tsuzuki & Yasuhiro Yoshioka. Jikken Igaku (Experimental Medicine), Volume 33, Issue 8, 2015, Pages 1273-1275. (in Japanese)